Many other government and private projects alike, including France’s highly anticipated ITER (International Thermonuclear Experimental Reactor), use tokamaks to heat up their star fuel. Even if new fission reactors can overcome these pitfalls, the energy source is still finite. The downside of this approach is that using radioactive atoms as the reactors’ energy source results in dangerous waste products and can also be susceptible to dangerous meltdowns like those that took place at Chernobyl in the 80s and Fukushima in 2011. 
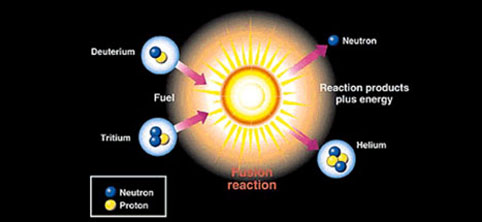
Depending on their size, nuclear power plants like these have an annual generating capacity of roughly 500-1000 megawatts, equivalent to up to the power of 1.3 million horses per plant. This energy is then used to heat water and create steam in order to turn turbines which create electricity. Nuclear fission is at the heart of nuclear power plants around the world and works by breaking apart the atoms of radioactive elements like uranium to create a burst of energy. While both reactions are nuclear, meaning they both involve energy changes at the center of atomic nuclei, they are actually polar opposites in many ways. M_\text$ "feels" maximum pull via strong force due to no one nucleon being "out of range" of another nucleon's pull.If nuclear fusion has so much potential, it may seem unclear why its counterpart, nuclear fission, can’t go toe-to-toe. The stronger the binding energy per nucleon, the less mass per nucleon.Įxample for neutron, proton and them bound together in deuterium: Mass defect is directly proportional to the binding energy. average energy per nucleus is higher before the reaction and lower after the reaction.Īfter the reaction - the resulting elements are closer to the iron atomic number The surplus energy is the energy taken out of the system, i.e. The surplus energy in the nuclear reaction is achieved when heavy elements are split (fission), or light element are fused (fusion). It is abundant in the universe, as natural atomic evolution tends to get close to it from both ends of atomic number spectrum. Both the lighter and heavier elements tend to have smaller binding energy. The literature presents the binding energy chart per element, with its peak at iron (~56 nucleons). Natural systems tend to evolve to lower, not higher energy states. The more energetic a binding, the more difficult it is to break the binding, the more stable the atom made out of such bindings. The more difficult it is to split the atom, the more stable the atom is. The higher the binding energy per nucleon, the more difficult it is to split the atom. The higher the binding energy of nucleus, the more energy is stored per nucleon in the system. The stronger the nucleus is bound, the higher its binding energy. Given (1) and (2), the smaller the nucleus, the stronger it is bound. The less nucleons in a nucleus, the closer in average they are, so the strong force per each is higher and easily overcomes electromagnetic repulsion. The more nucleons in a nucleus, the bigger the nucleus is, so the average distance of a nucleon to each another is higher, hence the long-distance electromagnetic repulsion tends to overcome short-distance strong nuclear force, up to the point of occasional alpha decay in elements of transuranic end of the spectrum. Please kindly point out and explain these errors. 
To me, each of them seems to make sense, but some of them are contradictory, so obviously - wrong. I did some research of that topic and cannot come to a single comprehensive and consistent description.īelow are related statements I gathered or can think of, describing the problem area. What is the nature of nuclear energy? This is closely related to the correct explanation of mass defect.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
